Eli Lilly Launches New Zepbound Pen: A Simpler Way to Manage Obesity
A redesigned monthly injection pen aims to simplify obesity treatment, improve adherence, and expand access to one of today’s most effective weight-loss therapies
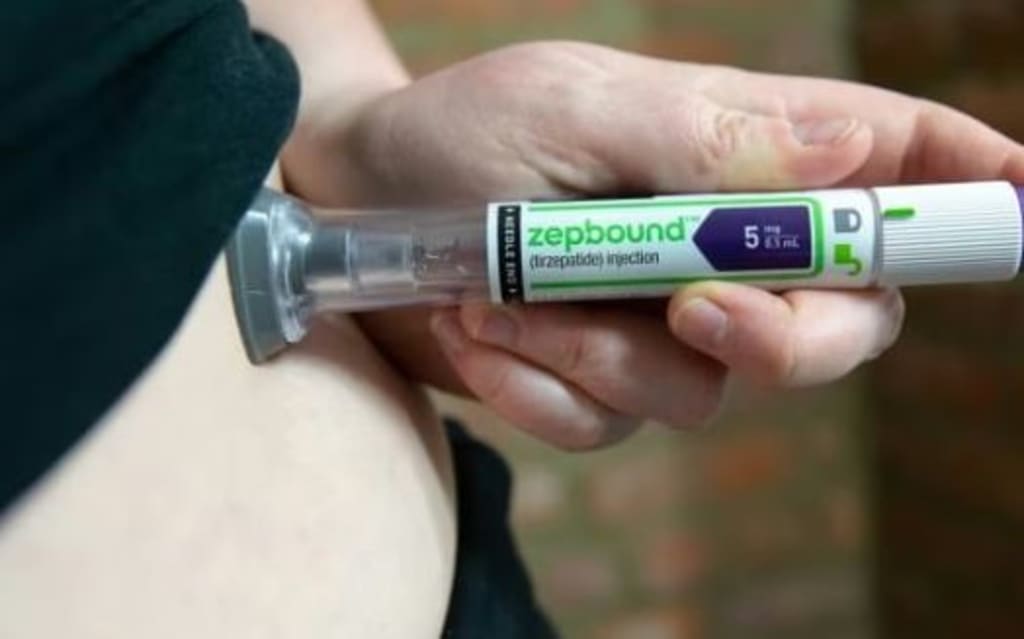
The fight against obesity is entering a new phase as Eli Lilly and Company unveils a redesigned version of its highly discussed weight-loss medication, Zepbound. The pharmaceutical giant has introduced a new delivery format that places an entire month’s worth of doses into a single injection pen — a move aimed at improving convenience, adherence, and accessibility for patients navigating long-term weight management.
The development reflects a broader trend in the pharmaceutical industry: making chronic medications easier to use. With obesity now recognized as a complex, long-term medical condition rather than simply a lifestyle issue, innovations in drug delivery are becoming almost as important as the medications themselves.
Why the New Pen Matters
Traditional injectable weight-loss medications often require patients to manage multiple pens, track dosing schedules carefully, and deal with storage challenges. Eli Lilly’s new single-pen design aims to streamline this experience.
Instead of juggling several weekly devices, patients can rely on one pen that covers four weeks of treatment. This reduces packaging waste, simplifies routines, and may lower the chances of missed doses — a key factor in treatment success.
For individuals who already manage other chronic conditions such as diabetes, fewer devices can mean less daily stress. Healthcare providers also benefit from easier patient education and potentially improved treatment adherence.
The launch highlights how pharmaceutical companies are increasingly focusing on user experience, not just clinical outcomes.
The Science Behind Zepbound
Zepbound belongs to a new class of medications that target hormones involved in appetite regulation and blood sugar control. By mimicking these hormones, the drug helps patients feel fuller for longer, reduce calorie intake, and improve metabolic health.
Clinical studies have shown significant weight reduction in many participants, positioning these therapies among the most effective non-surgical obesity treatments currently available. Researchers emphasize that the medication works best when combined with lifestyle changes such as balanced nutrition, physical activity, and behavioral support.
The shift toward hormone-based weight management represents a major scientific evolution. Rather than focusing solely on willpower, modern treatments acknowledge the biological mechanisms that influence hunger, metabolism, and body weight.
Convenience Could Improve Long-Term Success
One of the biggest challenges in obesity treatment is consistency. Weight management is not a short-term effort, and many patients struggle to stay on therapy due to complexity, cost, or frustration with routines.
The new multi-dose pen directly addresses this issue. A simpler system can reduce “treatment fatigue,” a phenomenon where patients gradually lose motivation to maintain complicated medication schedules.
From a psychological perspective, small improvements in convenience can have a meaningful impact. When treatment becomes easier, patients are more likely to stick with it — and sustained use is often necessary to maintain weight loss.
Healthcare professionals have long argued that adherence is one of the most overlooked factors in chronic disease outcomes. This innovation signals that drug makers are beginning to prioritize that reality.
Growing Competition in the Weight-Loss Market
Eli Lilly’s move also reflects intense competition in the rapidly expanding obesity drug market. Rival pharmaceutical firms are racing to develop similar therapies and delivery systems.
One of the most notable competitors is Novo Nordisk, which has seen massive demand for its own weight-loss medications. Supply shortages across the industry have highlighted just how strong consumer and clinical interest has become.
By improving device design, companies hope to differentiate their products beyond clinical results alone. Convenience, availability, insurance coverage, and long-term affordability are emerging as key battlegrounds.
Industry analysts predict that obesity therapeutics could become one of the largest pharmaceutical categories of the next decade, driven by rising global obesity rates and growing medical acceptance of drug-based treatment.
Regulatory Oversight and Safety
Any new drug format must pass regulatory review to ensure safety and effectiveness. Agencies such as the U.S. Food and Drug Administration evaluate both the medication itself and the delivery device.
Safety considerations include dosing accuracy, user instructions, device reliability, and potential side effects. Multi-dose pens must demonstrate that patients can use them correctly without increasing risk.
Regulators also monitor post-launch data to identify real-world outcomes. This ongoing oversight is particularly important for medications intended for long-term use.
Experts note that while these drugs show strong results, they are not suitable for everyone. Medical supervision remains essential, especially for patients with underlying health conditions.
A Shift in How Society Views Obesity
The popularity of medications like Zepbound reflects a broader cultural shift. Obesity is increasingly understood as a chronic disease influenced by genetics, environment, and physiology — not simply personal choices.
This shift has helped reduce stigma and opened the door for more comprehensive treatment strategies. Drug therapy is now often discussed alongside nutrition counseling, mental health support, and preventive care.
However, debates continue. Critics raise concerns about cost, access inequality, and whether long-term reliance on medication could overshadow lifestyle interventions. Supporters argue that effective medical tools are necessary to address a condition affecting hundreds of millions of people.
The new pen format fits into this evolving conversation by emphasizing practicality and sustainability in treatment.
What This Means for Patients
For patients considering weight-loss medication, the new design could remove some logistical barriers. Fewer devices, simpler routines, and potentially improved availability may make treatment more approachable.
Doctors may also find it easier to recommend therapy when patient management becomes less complicated. That could expand the number of people who benefit from clinically supervised weight management.
Still, experts stress that medication is not a standalone solution. Long-term success typically requires ongoing lifestyle changes, realistic expectations, and consistent medical follow-up.
Looking Ahead
Eli Lilly’s month-long Zepbound pen represents more than a packaging update — it signals how the obesity treatment landscape is maturing. As demand grows, innovation is moving beyond drug discovery into usability, supply, and patient experience.
Future developments may include oral versions, personalized dosing, digital tracking tools, and combination therapies targeting multiple metabolic pathways. The industry’s rapid pace suggests that today’s innovations are only the beginning.
For now, the new pen highlights a simple but powerful idea: when treatment becomes easier to manage, more people can stay on the path toward better health.





Comments
There are no comments for this story
Be the first to respond and start the conversation.