One Key Protein Could Be a Powerful New Target Against Malaria
Scientists identify a crucial malaria protein that could lead to powerful new treatments and vaccines. Targeting a single key protein may disrupt the parasite’s life cycle in humans and mosquitoes alike. Discovery of Aurora‑related kinase 1 offers hope for innovative antimalarial strategies. Researchers highlight a vulnerability in the malaria parasite that could transform global health interventions. New research points to a protein that could help combat drug-resistant malaria.
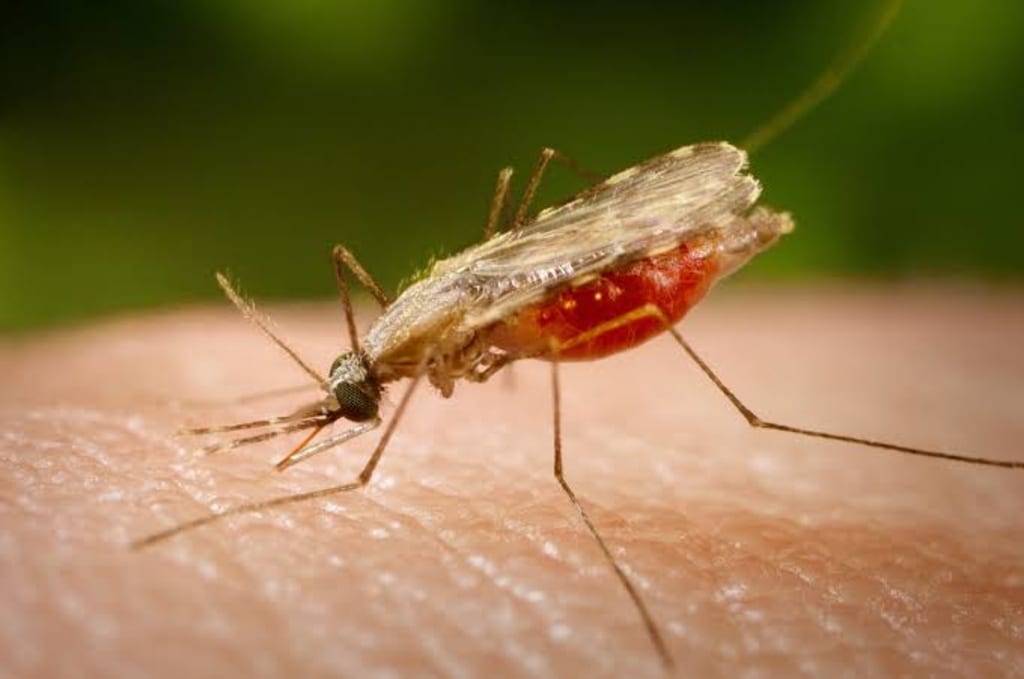
Scientists have uncovered a major vulnerability in the parasite that causes malaria that could lead to a new class of treatments and interventions, offering hope against one of the world’s deadliest infectious diseases. The discovery centers on a crucial protein called Aurora‑related kinase 1 (ARK1), which researchers say is indispensable to the malaria parasite’s ability to grow and reproduce.
Despite decades of progress in vaccine development and antimalarial drugs, malaria continues to kill hundreds of thousands of people each year, largely in sub‑Saharan Africa, with young children bearing the greatest burden. The need for new tools to combat the disease — especially ones that operate differently from existing drugs — is urgent. The newly identified protein may offer exactly that.
A Parasite’s Weakness Revealed
The malaria parasite, Plasmodium, undergoes a complex and unusual form of cell division that is very different from how human cells reproduce. ARK1 plays a central role in this process. The protein directs the assembly of a structure called the spindle apparatus, which ensures that the parasite’s genetic material is accurately split and distributed during cell division.
In laboratory experiments, scientists used genetic tools to turn off ARK1 in Plasmodium organisms. The result was dramatic: without this protein, the parasites could no longer form spindles correctly, meaning they failed to replicate at all — either inside human red blood cells or within the mosquito vectors that spread malaria. This effectively stopped their life cycle.
“This protein truly heralds a new beginning in our understanding of malaria cell biology,” said one researcher involved in the study. The finding was published in the scientific journal Nature Communications and represents a significant step forward in the fight against malaria.
Why ARK1 Is Such a Promising Target
There are two key reasons scientists are excited about ARK1 as a target:
1. Fundamental difference from human biology.
Humans have their own versions of Aurora kinases — proteins involved in cell division — but the form used by malaria parasites is structurally different. This means it may be possible to design drugs that specifically inhibit ARK1 in the parasite without interfering with human cells, reducing the risk of harmful side effects.
2. Blocking transmission as well as infection.
Most antimalarial drugs only affect the parasite once it has infected the human host. ARK1, however, is necessary both in the parasite’s human phase and in its reproductive stage inside mosquitoes. Targeting ARK1 could potentially disrupt the parasite’s life cycle in both hosts — a dual effect that would make new therapies more powerful than many existing options.
A New Direction for Antimalarial Therapy
Existing antimalarial drugs, such as artemisinin and its derivatives, often target metabolic processes within the parasite. Over time, however, resistance has emerged in some regions, reducing the effectiveness of these treatments. A drug targeting ARK1 could bypass these resistance mechanisms entirely, providing a novel mode of action.
Researchers emphasize that this discovery does not immediately translate into a new medicine. Drug development is a complex, costly, and time‑consuming process that involves designing molecules that effectively inhibit ARK1, testing them in laboratory and animal models, and eventually conducting clinical trials in humans. Nevertheless, identifying ARK1 as an essential protein is a major scientific breakthrough that opens the door to these next stages of research.
The Global Context
Malaria remains one of the most persistent global health challenges. According to health authorities, there were an estimated hundreds of millions of malaria cases in recent years, with a significant number of deaths — particularly among young children in areas lacking access to prompt treatment and preventive measures. While tools like bed nets, insecticides, and vaccines have helped reduce the burden, malaria has proved adept at evading control strategies through adaptation and resistance.
By identifying a previously underappreciated target like ARK1, scientists hope to stay ahead of the parasite’s evolutionary tricks. Future drugs that cripple the parasite’s ability to replicate could save countless lives and complement existing measures such as vaccines and vector control programs.
Looking Ahead
The discovery of ARK1’s critical role marks a promising chapter in antimalarial research. It underscores the value of fundamental biological studies in revealing “Achilles’ heels” within pathogens that can be exploited therapeutically. As researchers begin exploring molecules that can shut down this protein, the global health community may be on the cusp of a powerful new tool in the fight against one of humanity’s oldest foes.
About the Creator
Fiaz Ahmed
I am Fiaz Ahmed. I am a passionate writer. I love covering trending topics and breaking news. With a sharp eye for what’s happening around the world, and crafts timely and engaging stories that keep readers informed and updated.






Comments
There are no comments for this story
Be the first to respond and start the conversation.