What Molecular Weight Is GAPDH Protein?
Structure and Molecular Weight of the GAPDH Protein
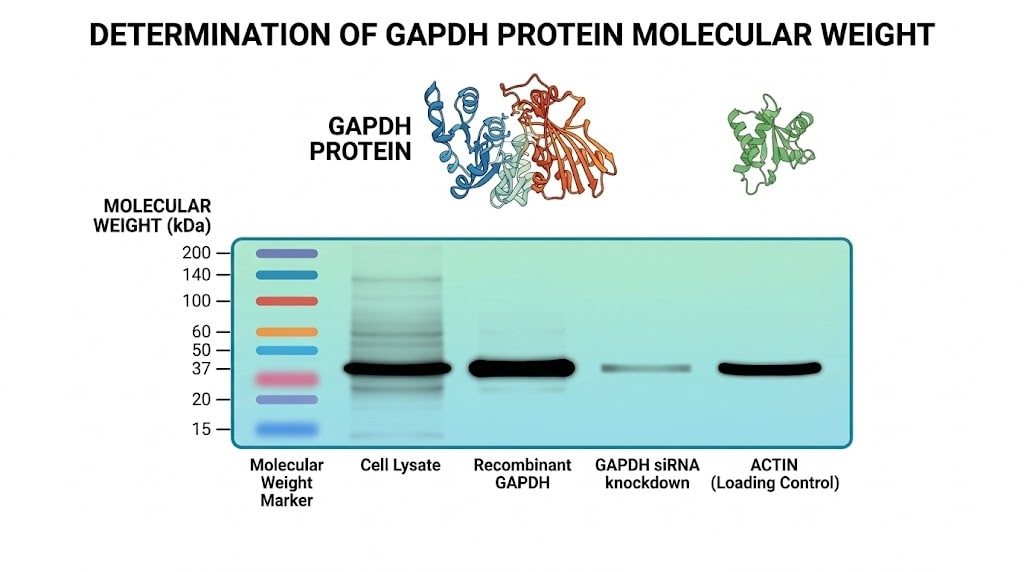
Understanding GAPDH kDa (kilodalton molecular weight) is fundamental for researchers using this protein as a loading control or housekeeping gene reference in molecular biology experiments. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) is a ubiquitously expressed enzyme with a predicted molecular weight of approximately 36-37 kDa. This consistent size and stable expression across cell types and experimental conditions have established GAPDH as one of the most widely used internal controls for normalizing protein expression in Western blotting and other quantitative techniques.
GAPDH Structure and Molecular Weight
The human GAPDH gene encodes a 335 amino acid protein with a calculated molecular mass of 36.05 kDa based on its primary sequence. However, on SDS-PAGE gels, GAPDH typically migrates as a band between 36-37 kDa, occasionally appearing slightly higher due to post-translational modifications or gel running conditions. The protein exists as a homotetramer in its active form, with each monomer contributing to the catalytic mechanism.
GAPDH's structure consists of two distinct domains: an NAD+ binding domain and a catalytic domain. The NAD+ binding domain contains a classic Rossmann fold that binds the nicotinamide adenine dinucleotide cofactor essential for catalytic activity. The catalytic domain contains the active site cysteine residue (Cys152) that forms a thiohemiacetal intermediate during the enzymatic reaction. This well-defined structure contributes to GAPDH's consistent molecular weight and stability across diverse biological samples.
Post-Translational Modifications
While the predicted GAPDH molecular weight is 36 kDa, post-translational modifications can slightly alter its migration on gels. GAPDH undergoes various modifications including phosphorylation, S-nitrosylation, acetylation, and ADP-ribosylation. These modifications generally do not substantially change the apparent molecular weight but may cause minor shifts in band position. Oxidative stress conditions can promote GAPDH aggregation or cross-linking, occasionally producing higher molecular weight species visible on Western blots.
GAPDH as Loading Control
GAPDH has become the gold standard loading control for Western blotting due to several advantageous properties. Its constitutive expression in virtually all cell types ensures presence across diverse sample types. The protein's abundance means researchers can detect it with short exposure times, and its stable expression under most experimental conditions makes it reliable for normalization. The well-defined 36-37 kDa molecular weight allows clear separation from many proteins of interest on standard gel percentages.
When using GAPDH as a loading control, researchers should verify that experimental treatments do not affect GAPDH expression levels. While generally stable, GAPDH can show altered expression under hypoxic conditions, certain drug treatments, or in specific disease states. Including multiple loading controls or using total protein staining as an additional normalization method helps ensure accurate quantification.
Optimal Detection Conditions
For reliable GAPDH detection at 36-37 kDa, researchers should optimize gel percentage and running conditions. Standard 10-12% acrylamide gels provide excellent resolution in the 30-40 kDa range. Using molecular weight markers that span this range enables accurate band identification. GAPDH antibodies work effectively at relatively high dilutions due to the protein's abundance—typical dilutions range from 1:5,000 to 1:20,000 depending on antibody quality and sample type.
Alternative Housekeeping Proteins
While GAPDH remains widely used, researchers should consider alternative loading controls depending on experimental context. Beta-actin (42 kDa), alpha-tubulin (50 kDa), and beta-tubulin (50 kDa) offer different molecular weights that may better separate from target proteins. Histone H3 (17 kDa) serves well for nuclear protein studies. Using loading controls with molecular weights distinct from proteins of interest prevents band overlap and facilitates accurate quantification.
The choice between GAPDH and alternative housekeeping proteins depends on several factors. If the protein of interest migrates near 36-37 kDa, selecting a loading control with a different molecular weight prevents confusion. For experiments where GAPDH expression might change (hypoxia studies, certain cancer research), alternative controls provide more reliable normalization. Validating multiple housekeeping proteins for experimental conditions ensures choosing the most stable reference.
Species Differences
GAPDH is highly conserved across species, with molecular weights remaining remarkably consistent. Human, mouse, rat, and other mammalian GAPDH proteins all migrate around 36-37 kDa. This conservation enables using the same antibodies across multiple species—many GAPDH antibodies demonstrate cross-reactivity with human, mouse, rat, and other vertebrate species. Researchers working with multiple species can confidently expect GAPDH to appear at the same molecular weight position across samples.
Troubleshooting GAPDH Detection
If GAPDH fails to appear at the expected 36-37 kDa position, several factors may be responsible. Degraded samples produce lower molecular weight bands as proteases cleave GAPDH. Incomplete denaturation or reduction can cause aberrant migration. Running gels at improper percentages may compress or expand the separation range, shifting band positions. Using fresh samples, ensuring complete protein denaturation with appropriate sample buffers, and selecting proper gel percentages typically resolves these issues.
Multiple GAPDH bands sometimes appear due to post-translational modifications, incomplete denaturation causing partial oligomerization, or cross-reactivity with related proteins. Including appropriate positive controls from well-characterized cell lysates helps distinguish genuine GAPDH signals from artifacts. When multiple bands appear, the strongest band at 36-37 kDa typically represents the primary GAPDH species suitable for normalization.
Conclusion
GAPDH's molecular weight of 36-37 kDa makes it an ideal loading control for Western blotting and other protein quantification techniques. Its consistent size, ubiquitous expression, and stable levels under most experimental conditions have established GAPDH as a cornerstone of molecular biology research. Understanding the expected molecular weight and factors that might influence it enables researchers to use GAPDH effectively for normalizing protein expression data.
Researchers should validate GAPDH stability in their specific experimental systems, consider alternative loading controls when appropriate, and employ proper detection methods to ensure accurate quantification. With careful attention to technical details and appropriate controls, GAPDH remains a reliable and convenient tool for normalizing protein expression across diverse research applications.
About the Creator
Mia Bella
A beauty enthusiast and expert, Mia Bella is dedicated to sharing the latest trends, tips, and product reviews. With a passion for skincare and makeup, she empower others to enhance their natural beauty and confidence.






Comments